 Chronic inflammation is a prominent feature of obesity and a major cause of insulin resistance ( 9). Nevertheless, an important issue concerning the potential links between metabolic inflammation, nitrosative stress, and declined hepatic autophagy remains unsolved. These observations implicate GSNOR as a key regulator of hepatic autophagy and insulin action in obesity and argue that boosting GSNOR activity may exert a metabolic benefit due to its role in sustaining hepatic autophagy. If, however, those mice expressed a GSNOR transgene in their livers, hepatic autophagy was restored and glucose homeostasis and insulin sensitivity improved significantly ( 5). In obese mice, GSNOR defect exacerbated the downregulation of hepatic autophagy and impaired hepatic insulin action. ( 5) is that obesity hampers the denitrosylation capacity of the liver, which appears to be a major cause of impaired hepatic autophagy and insulin resistance under the obese condition. Denitrosylation enzymes, particularly S-nitrosoglutathione reductase (GSNOR) and thioredoxin, are critical modulators of protein S-nitrosylation ( 8). NO regulates a range of cellular functions and signaling processes through protein S-nitrosylation, the covalent attachment of a nitrogen monoxide group to the thiol side chain of cysteine, by altering protein subcellular localization, enzyme activity, protein stability, and protein complex formation ( 7). Indeed, hyper–NO modification of HexB and CTSB was detected in livers of human patients with diabetes or patients with advanced hepatic steatosis ( 5). 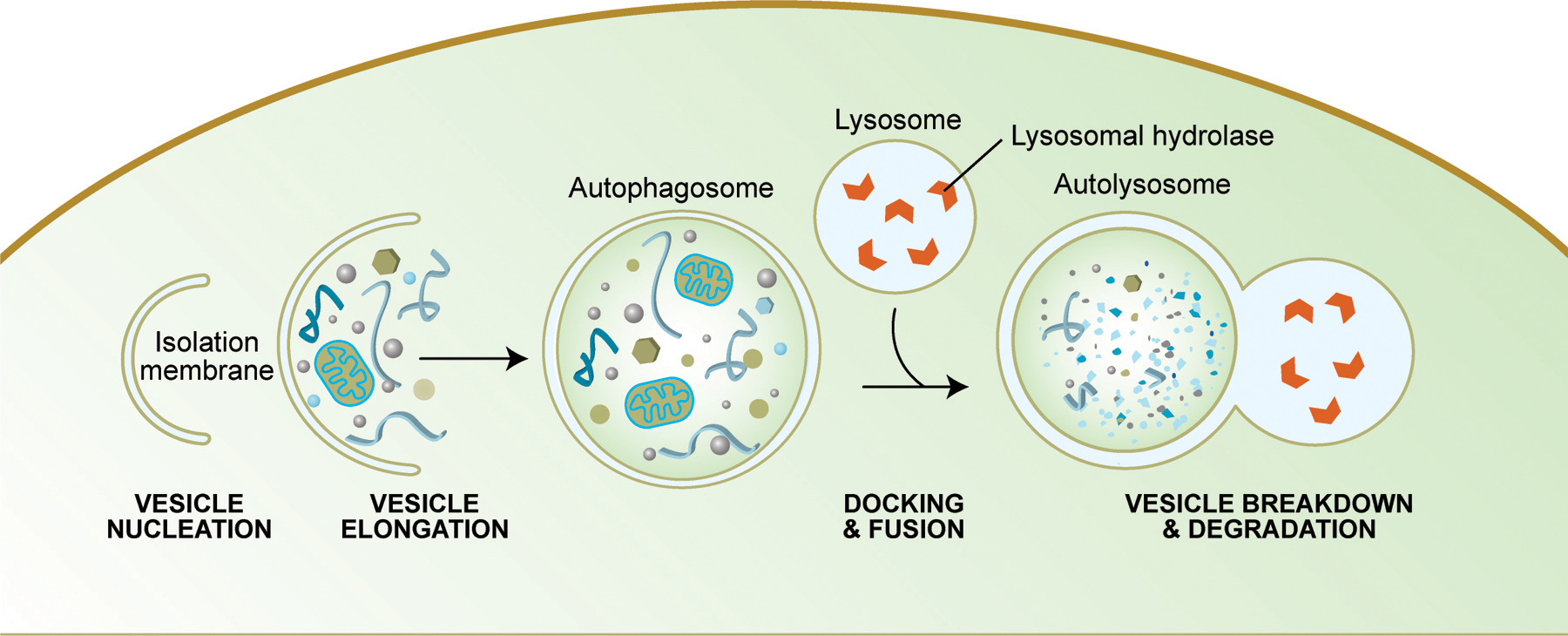
In particular, obesity increased S-nitrosylation of two lysosomal enzymes, cathepsin B (CTSB) and hexosaminidase subunit β (HexB), thus impairing lysosomal function ( Fig. Their study shows that diet-induced obesity triggers a global increase in protein S-nitrosylation in mouse livers. provided novel insights into the mechanistic underpinning of impaired hepatic autophagy in obesity. Compared with the earlier studies, Qian et al. ( 5) confirmed that hepatic autophagic insufficiency compromises the liver’s ability to adapt to metabolic stress and thereby cripples hepatic insulin action. Declined autophagic activity is associated with hepatic steatosis and insulin resistance in obesity, where, on one hand, insulin resistance downregulates expression of the genes encoding major autophagy components ( 6) while, on the other hand, impaired autophagy promotes insulin resistance by exacerbating hepatic steatosis ( 4). In the liver, autophagy participates in the basal turnover of lipids by engulfing and degrading lipid droplets ( 3). 1).Īutophagy is an adaptive, catabolic process that generates energy for cells under nutrient-starvation conditions and helps maintain cellular homeostasis in nutrient-rich environments through its constitutive activity ( 2). Their data argue that in obese animals, NO-induced repression of hepatic autophagy causes hepatic steatosis, impairs hepatic insulin signaling, and eventually contributes to the progression of type 2 diabetes ( Fig. ( 5) revealed a mechanism by which obesity cripples autophagy in the liver through S-nitrosylation, an inhibitory protein modification process induced by nitric oxide (NO). In this issue of Diabetes, a study conducted by Qian et al. 
However, the mechanisms underlying the cause of autophagic defect in metabolic disorders remain elusive. 
Dysfunction of autophagy is associated with obesity and type 2 diabetes. Autophagy, a lysosomal degradation pathway for damaged organelles or long-lived proteins, was recently identified as a key regulatory pathway to preserve lipid homeostasis and insulin sensitivity under metabolic stress conditions ( 2– 4). Intensive research efforts have linked chronic inflammation, redox changes, and intracellular stress responses to dysregulated energy metabolism, particularly insulin resistance ( 1). During the progression from obesity to diabetes, a critical attribute is inability to maintain metabolic homeostasis under excessive energy and nutrient exposure, which triggers insulin resistance. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
